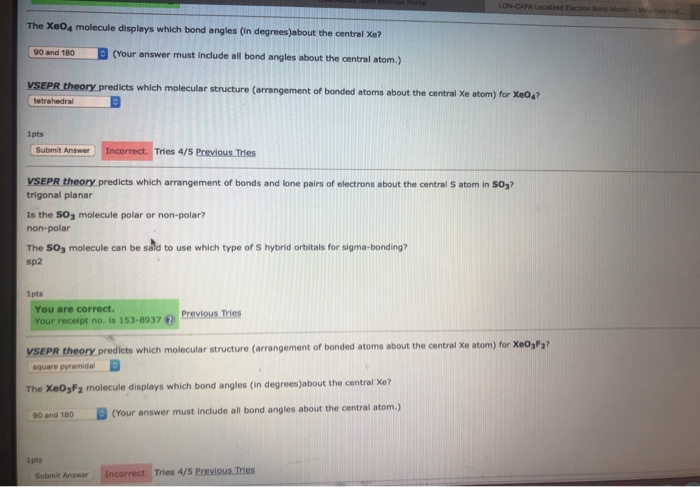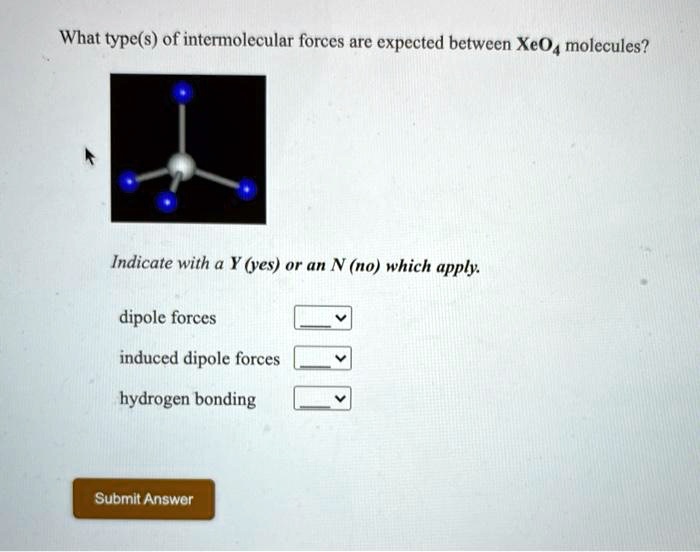
SOLVED: What type(s) of intermolecular forces are expected between XeO4 molecules? Indicate with a Y (yes) or an N (no) which apply: dipole forces induced dipole forces hydrogen bonding Submit Answor

SOLVED: Lewis Structures Draw the Lewis structures for the following compounds Make certain that: Show all calculations for building up and using the valence electron pool. b. All atoms have their correct

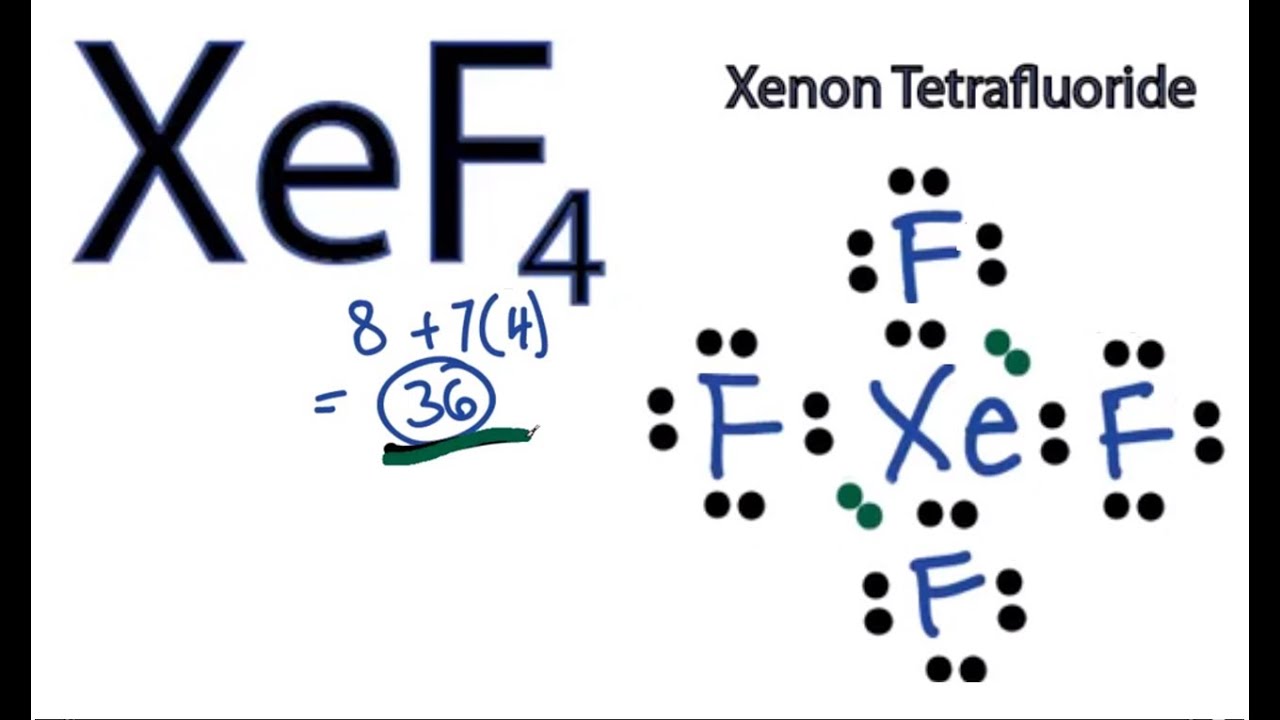
Draw the Lewis structure for XeO_4 and then decide if the molecule is polar or nonpolar. | Homework.Study.com