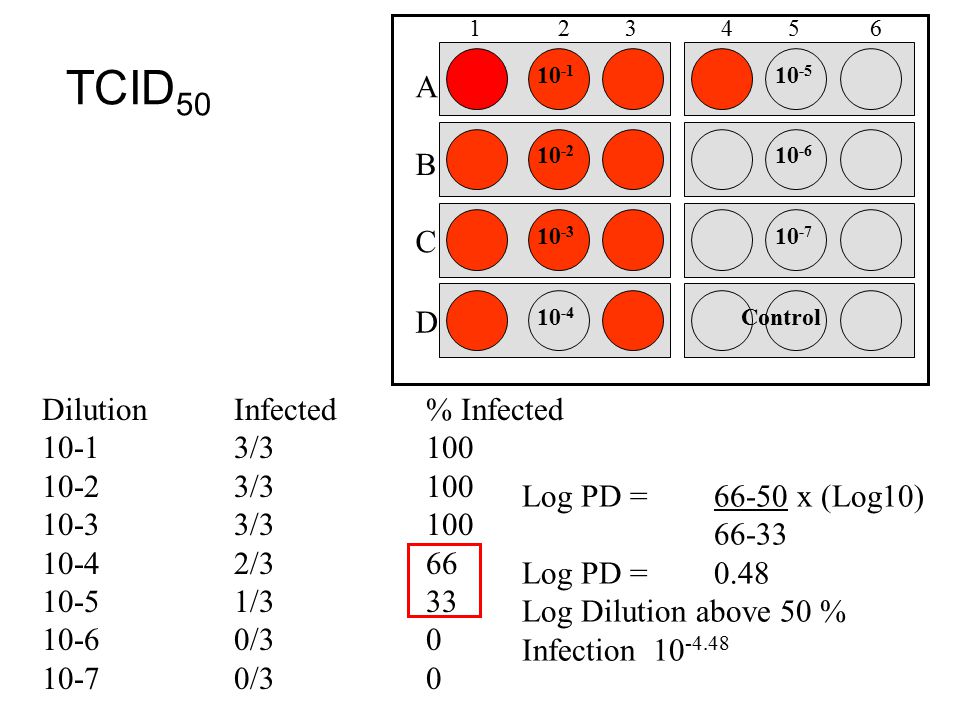
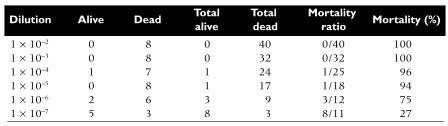
www.bio-protocol.org/e2855 Virucidal and Neutralizing Activity Tests for Antiviral Substances and Antibodies Chie Aoki-Utsubo1,
Time to revisit the endpoint dilution assay and to replace the TCID50 as a measure of a virus sample's infection concentration | PLOS Computational Biology
Rapid Titration of Measles and Other Viruses: Optimization with Determination of Replication Cycle Length | PLOS ONE

Improvements in methods for calculating virus titer estimates from TCID50 and plaque assays. | Semantic Scholar
Evaluation of SARSâ•'CoVâ•'2 neutralizing antibodies using a CPEâ•'based colorimetric live virus microâ•'neu
Relationship between hemagglutinin stability and influenza virus persistence after exposure to low pH or supraphysiological heating | PLOS Pathogens
Supplementary Materials Materials and Methods Virucidal suspension test. MRC-5 cells were maintained as monolayers in disposable

![TCID50 Titer Calculation adapted from [3] | Download Scientific Diagram TCID50 Titer Calculation adapted from [3] | Download Scientific Diagram](https://www.researchgate.net/profile/Paul-Lemieux-3/publication/317720939/figure/fig8/AS:507677378912265@1498051127683/TCID50-Titer-Calculation-adapted-from-3.png)
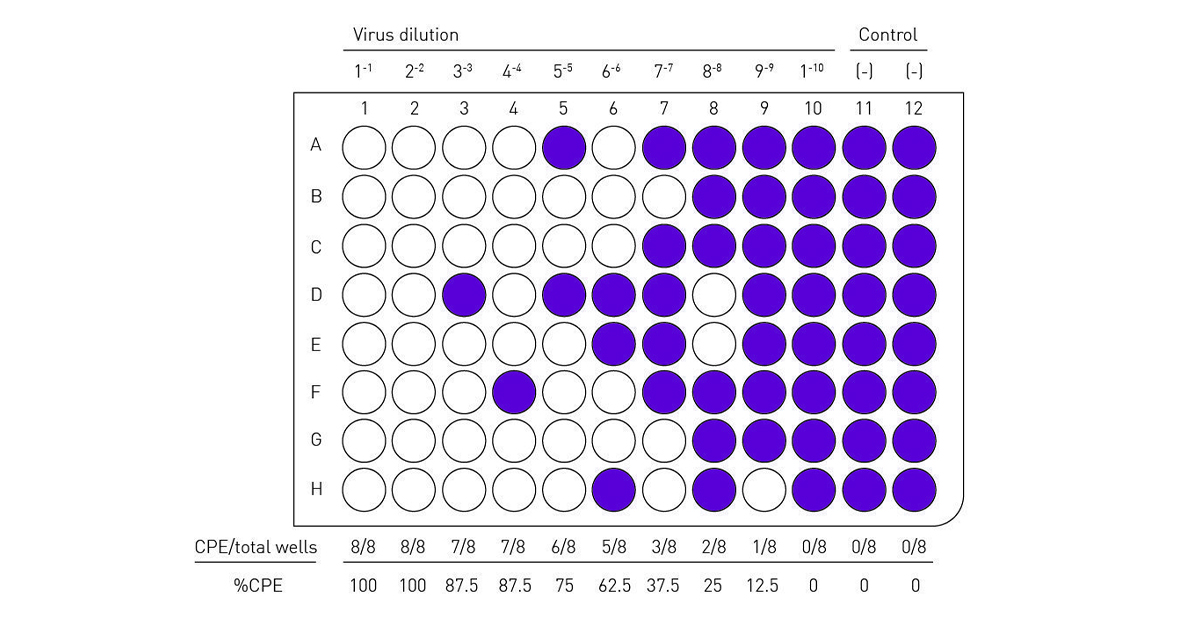




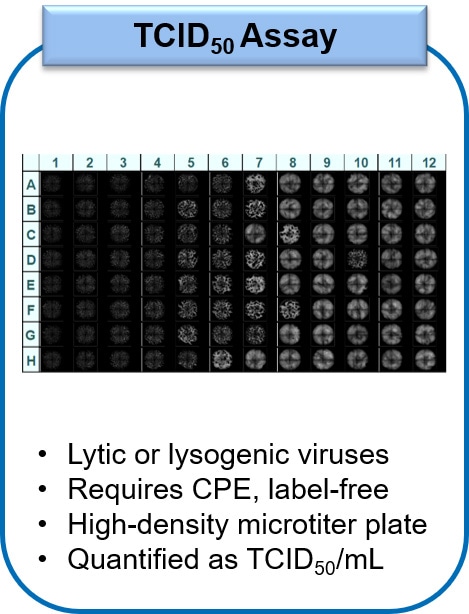
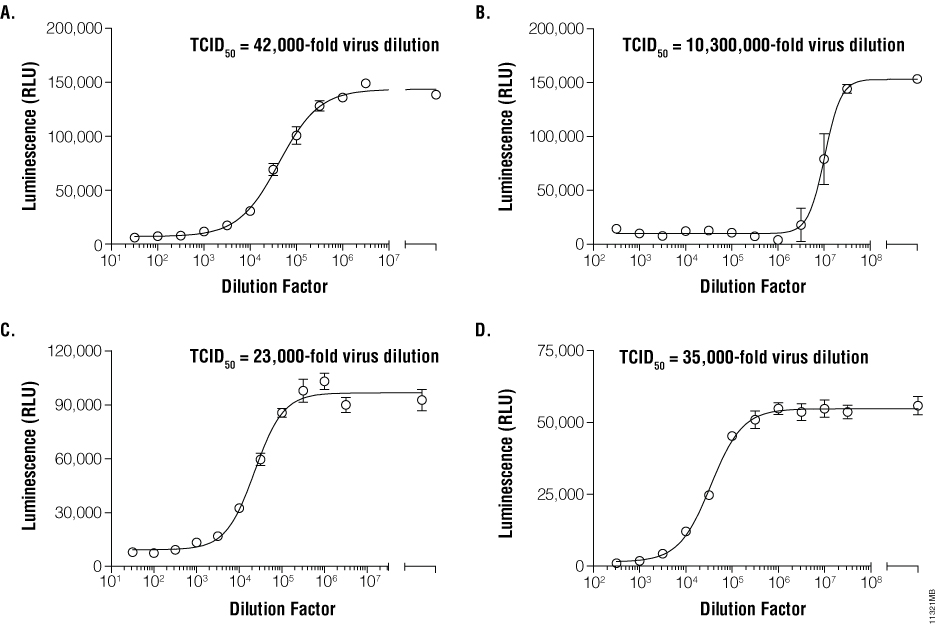
![TCID50 Titer Calculation adapted from [3] | Download Scientific Diagram TCID50 Titer Calculation adapted from [3] | Download Scientific Diagram](https://www.researchgate.net/profile/Paul-Lemieux-3/publication/317720939/figure/fig8/AS:507677378912265@1498051127683/TCID50-Titer-Calculation-adapted-from-3_Q320.jpg)