Content Uniformity Testing Through Utilization of Automated Dissolution Technologies | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology
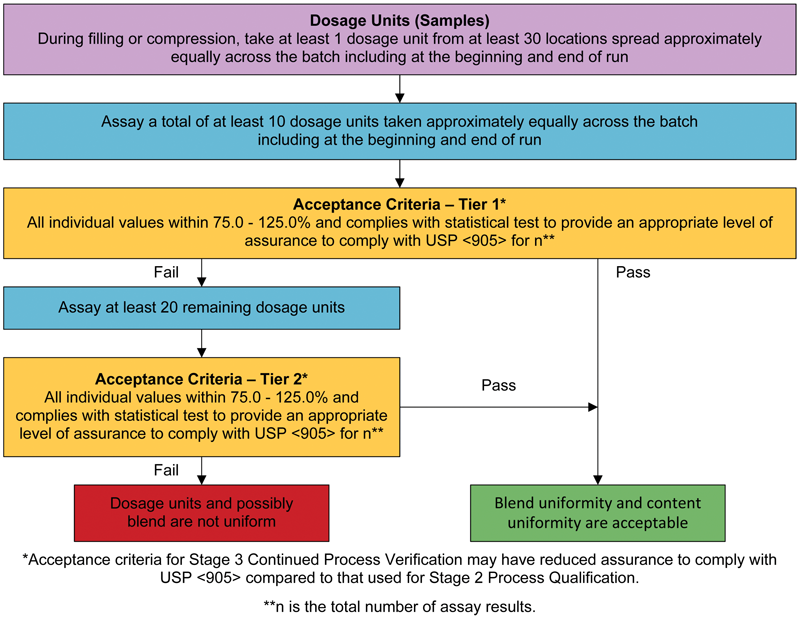
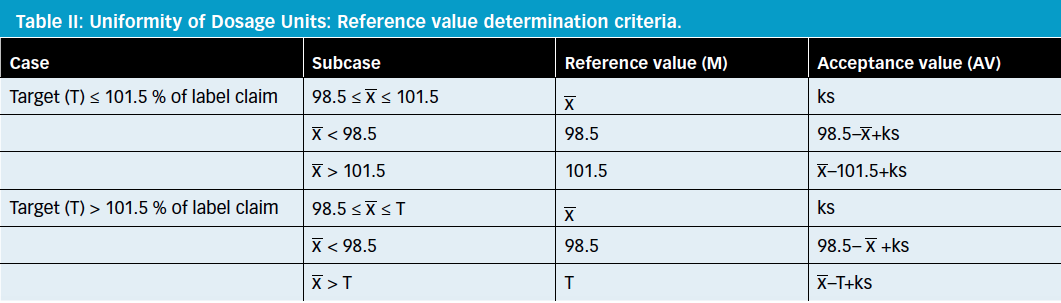
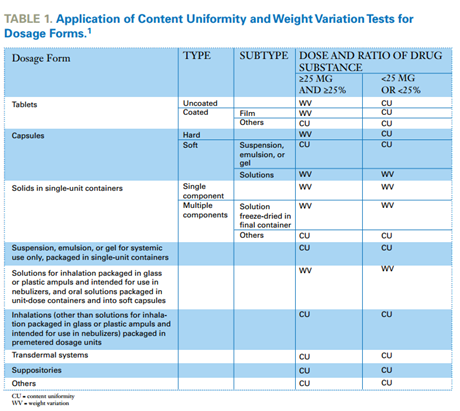
Comparison of Two Relevant Statistical Approaches to Assess Content Uniformity | Pharmaceutical Engineering

Raman spectroscopy as a complementary tool to assess the content uniformity of dosage units in break-scored warfarin tablets - ScienceDirect

The study of the applicability of content uniformity and weight variation test--the state of commercial tablets and capsules in Japan. | Semantic Scholar
Comparison of Two Relevant Statistical Approaches to Assess Content Uniformity | Pharmaceutical Engineering
![PDF] Weight and content uniformity of lorazepam half-tablets: A study of correlation of a low drug content product. | Semantic Scholar PDF] Weight and content uniformity of lorazepam half-tablets: A study of correlation of a low drug content product. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3f129f060436923435cbb5e514e797c1b6799cb1/3-Table2-1.png)