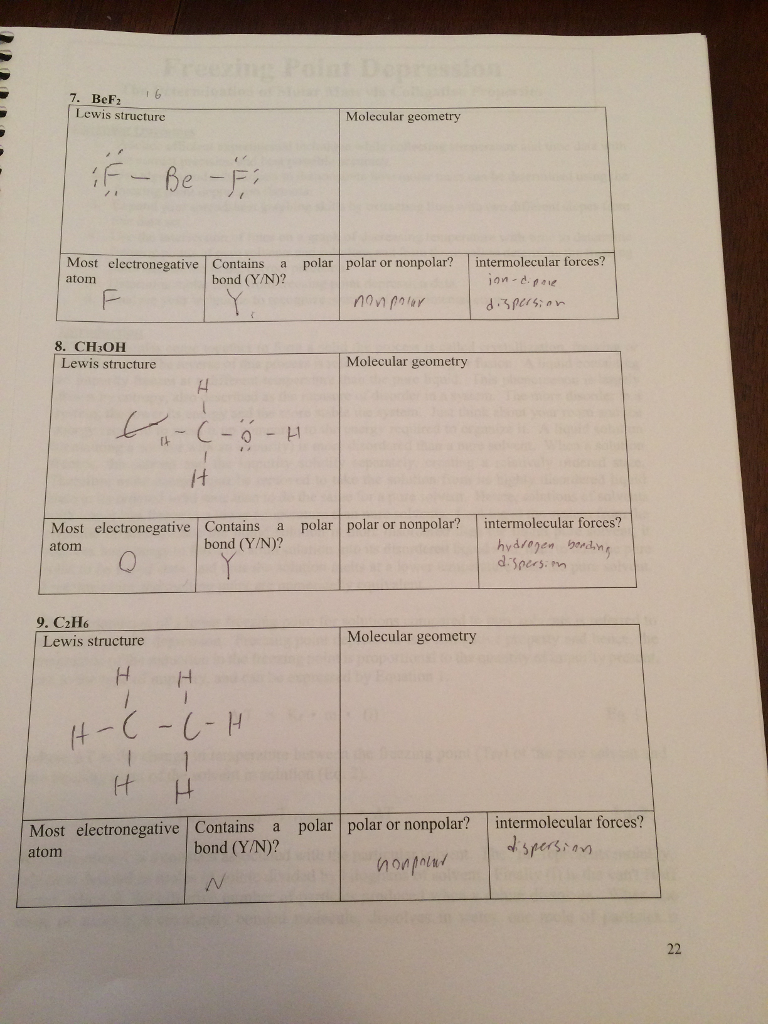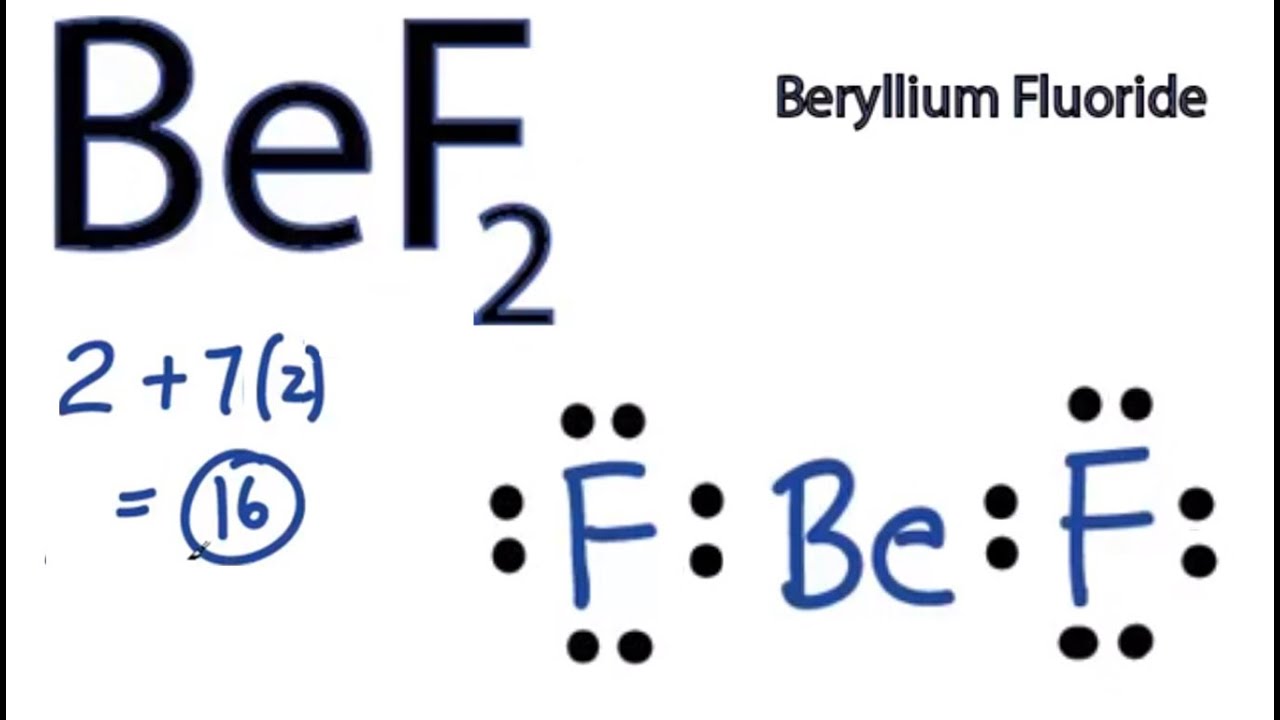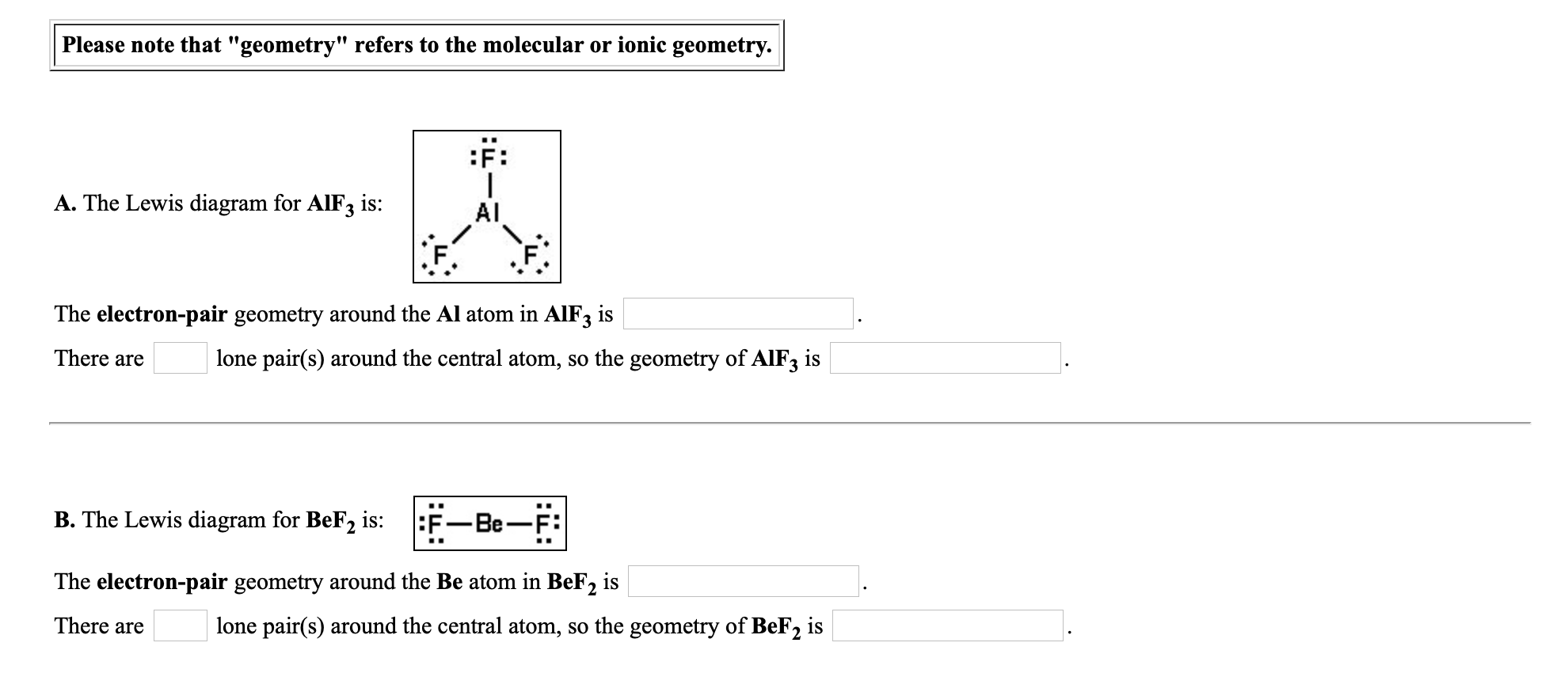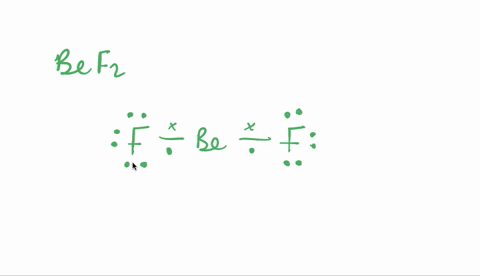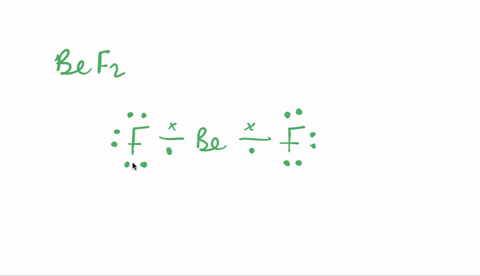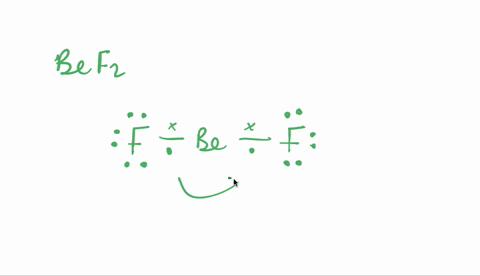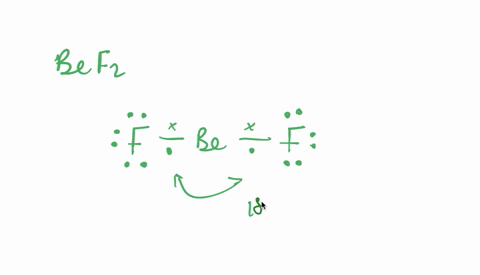
SOLVED:Despite the large electronegativity difference between fluorine and beryllium atoms, BeF2 forms linear molecules in gas phase. Are these molecules polar or nonpolar? Explain.

BeF2 Lewis Structure (Beryllium Fluoride) | BeF2 Lewis Structure (Beryllium Fluoride) BeF2 is a chemical formula for Beryllium fluoride. It consists of one Beryllium and two Fluorine atoms. In... | By Geometry

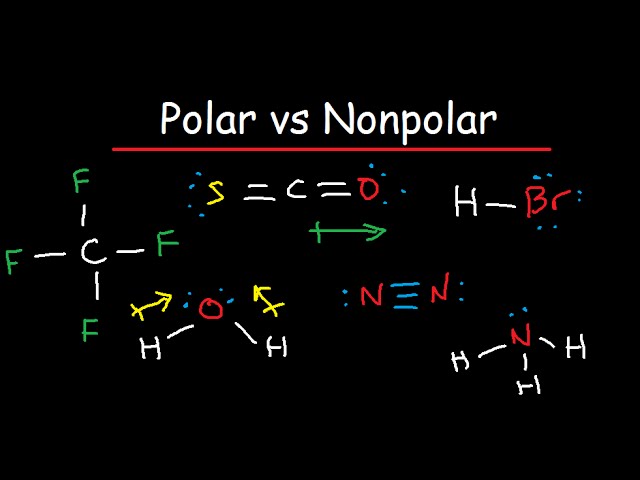
UNIT IX Lesson #2 Polar Vs Non Polar. Review BOND TYPEWHAT TO LOOK FOR... INTRAMOLECULAR BONDS ( within the molecule itself) IONIC BONDMade up of metal. - ppt download
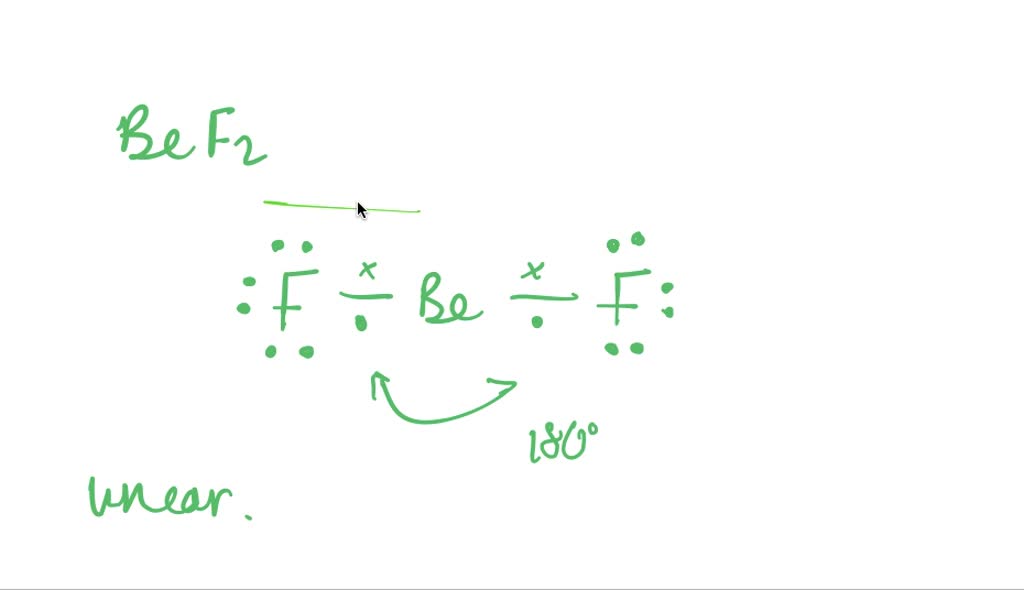
SOLVED:Despite the large electronegativity difference between fluorine and beryllium atoms, BeF2 forms linear molecules in gas phase. Are these molecules polar or nonpolar? Explain.